Electrochemistry (12th) MockTest (Chemistry) MHT CET 2021
Test Instructions:
- This Mock test contains 15 MCQ questions.
- Choice and sequence for attempting questions will be as per the convenience of the candidate.
- Read each question carefully.
- Determine the one correct answer out of the four available options given for each question.
- Each question awards you 1 point for each question.
- You can end the test any time by clicking “quiz summary>finish quiz”.
- The mock test will automatically be submitted when the time is up.
- You can review all questions once again and evaluate your mistakes by clicking “view questions” when the result is displayed.
- You can apply for Leaderboard when the result is displayed.
- You can re-attempt the exam as per your wish, yet scores of the first attempt will be recorded!
Test Summary
0 of 15 Questions completed
Questions:
Information
|
You must fill out this field. |
You have already completed the test before. Hence you can not start it again.
Test is loading…
You must sign in or sign up to start the test.
You must first complete the following:
Results
Results
0 of 15 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
| Pos. | Name | Entered on | Points | Result |
|---|---|---|---|---|
| Table is loading | ||||
| No data available | ||||
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 15
1. Question
1 point(s)The unit of molar conductivity is ______.
-
Question 2 of 15
2. Question
1 point(s) -
Question 3 of 15
3. Question
1 point(s)The charge of one mole electrons is
-
Question 4 of 15
4. Question
1 point(s)The atomic weight of Fe is 56 u. The weight of Fe deposited from FeCl3 solution by passing 0.6 faraday of electricity is _______.
-
Question 5 of 15
5. Question
1 point(s) -
Question 6 of 15
6. Question
1 point(s)Nernst equation for the following cell reaction at 25℃ is :

-
Question 7 of 15
7. Question
1 point(s)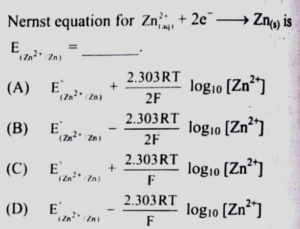
-
Question 8 of 15
8. Question
1 point(s)Standard hydrogen electrode _____.
-
Question 9 of 15
9. Question
1 point(s)In H2 – O2 fuel cell the electrodes are made up of _______.
-
Question 10 of 15
10. Question
1 point(s)
-
Question 11 of 15
11. Question
1 point(s)The number of electrons required to reduce
4.5 x 10-5 g of aluminium is ______.
-
Question 12 of 15
12. Question
1 point(s)For how much time, 10 A electric current should be passed through a dilute aqueous NiSO4 solution during electrolysis using inert electrode, in order to get 5.85 g Nickel ?
[At. mass of Ni = 58.5 u]
-
Question 13 of 15
13. Question
1 point(s)
-
Question 14 of 15
14. Question
1 point(s)For the following cell , reaction at cathode is :

-
Question 15 of 15
15. Question
1 point(s)Which of the following is INCORRECT about mercury cell ?
